test
Enarthronota Grandjean, 1969
Superorder Acariformes » Order Sarcoptiformes » Suborder Oribatida » Infraorder Enarthronota
superfamilies: Atopochthonioidea, Brachychthonioidea, Heterochthonioidea, Hypochthonioidea, Protoplophoroidea
enarthronote oribatid mites
low
No known quarantine importance. Most species are small to minute fungivores in mostly dry soils, including house dust and rarely stored products.
White to yellow, tan, brown, or rarely more brightly coloured armoured oribatid mites with 1–3 hysterosomal scissures, or their remnants, or pytchoidy. Pedofossae sometimes present. Bodies rectangular, elongate, oval or globular; opisthosomal glandsopisthosomal glands:
see oil glands.
absent. Setaeseta:
(pl. setae, from L. = bristle) cuticular process composed of a hollow shaft (sometimes filled with a refractive material) produced from a membranous socket (the alveolus); the hair-like, spine-like, branched or variously expanded structures on the surfaces of the legs and body. Most setae function as mechanoreceptors, but others (e.g., solenidia) are chemoreceptors or have unknown or ambiguous functions. ranging from setiform, to brush-like, to highly modified leaf-like shapes or ornate dendritic arrangements. Capitulumgnathosoma:
ranging from setiform, to brush-like, to highly modified leaf-like shapes or ornate dendritic arrangements. Capitulumgnathosoma:
(= capitulum) the anteriormost part of a mite or ricinuleid, composed of the cheliceral and pedipalpal segments and separated from the body (idiosoma) by a ring of soft cuticle.
usually withdrawn into a camerostomecamerostome:
a recess under the rostral tectum that allows retraction of the chelicerae and palps of oribatid mites and that is sealed by the subcapitulum when retracted; a deep recess containing the gnathosoma in Uropodina.; chelicerae sometimes visible from above; a pair of lateral eyes rarely present. Macropyline, 3 pairs of genital papillaegenital papillae:
1-3 pairs of extrusible finger-like to button-like projections, usually retracted into in the genital vestibule of acariform mites; sometimes formed as sessile disks around the genital opening; thought to be osmoregulatory structures; modified or multiplied and dispersed over the body in many freshwater mites. Genital papillae are absent in the larva, but may be added ontogenetically: protonymphs have one pair, deutonymphs two pairs, and tritonymphs (and adults) three pairs. The tritonymphal pair of papillae is often lost. The serially homologous Claparède's organ is usually present in the larvae (and prelarvae) of mites exhibiting genital papillae in nymphs and adults (Oudeman's Rule)..
Some Endeostigmata somewhat resemble enarthronotes, but are soft-bodied and lack scissures (although remnants of primary segmentation may be present).
Fossil enarthronotes are known from the Devonian. Extant taxa are found in most soil types, but are especially abundant in the drier soils and in mosses. Many species are all female parthenogens. The feeding ecology of this group is poorly understood, but many have highly modified chelicerae, cheliceral setaeseta:
(pl. setae, from L. = bristle) cuticular process composed of a hollow shaft (sometimes filled with a refractive material) produced from a membranous socket (the alveolus); the hair-like, spine-like, branched or variously expanded structures on the surfaces of the legs and body. Most setae function as mechanoreceptors, but others (e.g., solenidia) are chemoreceptors or have unknown or ambiguous functions. , and adoral setaeseta:
, and adoral setaeseta:
(pl. setae, from L. = bristle) cuticular process composed of a hollow shaft (sometimes filled with a refractive material) produced from a membranous socket (the alveolus); the hair-like, spine-like, branched or variously expanded structures on the surfaces of the legs and body. Most setae function as mechanoreceptors, but others (e.g., solenidia) are chemoreceptors or have unknown or ambiguous functions. . Brachychthonioids are found everywhere, usually in high diversity, feed on algae and other microbes, and are among the smallest of all oribatid mites. Hypochthonioids are generally dorsoventrally flattened, but Mesoplophoridae are globular, lack any scissures, and exhibit ptychoidyptychoidy:
. Brachychthonioids are found everywhere, usually in high diversity, feed on algae and other microbes, and are among the smallest of all oribatid mites. Hypochthonioids are generally dorsoventrally flattened, but Mesoplophoridae are globular, lack any scissures, and exhibit ptychoidyptychoidy:
the ability of some oribatid mites to withdraw the legs between two body regions like a penknife being closed or a box being closed (hence box mites), and resulting in a seed-like appearance. A derived form of dichoidy.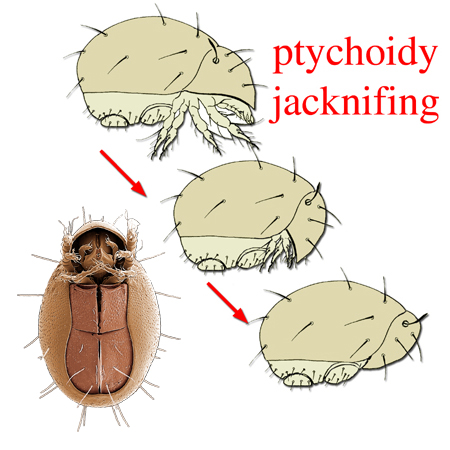 . Protoplophoroids range from poorly sclerotised endeostigmatanendeostigmatan:
. Protoplophoroids range from poorly sclerotised endeostigmatanendeostigmatan:
a member of the Endeostigmata; characteristic of or belonging to the Endeostigmata.
-like mites (e.g., Paralycus), to rather brachythonioid-like mites (e.g., Haplochthonius), to strongly ornamented mites with elongate erectile setaeseta:
(pl. setae, from L. = bristle) cuticular process composed of a hollow shaft (sometimes filled with a refractive material) produced from a membranous socket (the alveolus); the hair-like, spine-like, branched or variously expanded structures on the surfaces of the legs and body. Most setae function as mechanoreceptors, but others (e.g., solenidia) are chemoreceptors or have unknown or ambiguous functions. (e.g., Cosmochthonius), to light-bulb shaped subsub:
(e.g., Cosmochthonius), to light-bulb shaped subsub:
- as prefix used to indicate (1) under (see subcapitulum) or (2) not quite, e.g., subrectangular = not quite rectangular; subtriangular = not cleanly triangular.
-ptychoid mites (e.g., Sphaerochthonius), to fully ptychoid taxa (e.g., Protoplophora).