NOCTUIDAE - Spodoptera litura (Fabricius)
Taxonomy
Noctuoidea: Noctuidae: Noctuinae: Spodoptera litura (Fabricius)
Common names: rice cutworm, cluster caterpillar, cotton leafworm, tobacco cutworm, tropical armyworm, Egyptian cottonworm
Synonyms: Prodenia litura, Noctua histrionica, Noctua elata, Prodenia ciligera, Prodenia tasmanica, Prodenia subterminalis, Prodenia glaucistriga,
Prodenia declinata, Mamestra albisparsa, Prodenia evanescens, Orthosia conjuncta
Larval diagnosis (Summary)
- Mandible with scissorial teeth resulting in a serrate cutting edge
- Ground color green to yellow brown to dark blue gray
- Subdorsal area often not contrasting with paler dorsum
- Middorsal line often present and conspicuous
- Spiracular stripe, if interrupted on A1, then equal in intensity on both the thorax and abdomen
- Dorsal triangles, if present, usually with an apical white dot
- Abdominal spiracles usually with a large black dot dorsally and a white spot posteriorly
- From Middle East to Asia on a wide range of hosts
Host/origin information
More than 85% of all interception records at U.S. ports of entry for S. litura are from Thailand on orchids.
| Origin |
Host(s) |
| Thailand | Dendrobium, Oncidium |
| Malaysia | various |
| Singapore | various |
Recorded distribution
Spodoptera litura is widely distributed throughout Asia and Australasia, from Afghanistan, northwestern India, and Pakistan to Korea, China, and Japan,
south to Australia and New Zealand. It is also present on many Pacific Islands as well as in Hawaii (Pogue 2002).
Identification authority (Summary)
Because the genus Spodoptera is hard to define as larvae, care must be taken to be sure the specimen is in this genus.
Detailed descriptions of morphology and coloration are included on the Detailed Information page.
Because this pest is polyphagous, the hostplant offers few clues. The one exception is S. litura on orchids from Thailand.
Pest characterization
(Based on Cavey 2001, Pogue 2002)
- Taxonomy: High. Species identification is possible in late instars.
- Distribution: High. Spodoptera litura does not occur in the U.S.
- Potential Impact: High. Spodoptera litura is a serious pest.
This ranking characterizes Spodoptera litura as quarantine significant for the U.S.
NOCTUIDAE - Spodoptera litura (Fabricius)
Larval diagnosis (Detailed)
The larva of S. litura, usually called the rice cutworm (USDA 1982), was described by Chu et al. (1965), Thakar and Srivastva (1983) and
partially described by Gardner (1941, 1947), Mochida (1973), Weisman (1974), Bejakovich and Dugdale (1998), Pogue (2002), Sri et al. (2010), and Passoa (2011).
Srivastava and Mathur (1964) described the mouthparts. Cox (1976), Bejakovich and Dugdale (1998), Sugi (1987), Wagner et al. (2011), and
Komai et al. (2011) illustrated the larva in color.
Weisman (1986) did not include S. litura in his key. Several authors have partially described the larva, usually emphasizing different character sets.
Sometimes there is conflicting information. Due to a lack of a modern detailed morphological description, we review the major available literature
on the larval morphology of S. litura to support our diagnosis of this important quarantine pest.
Gardner (1941) studied S. litura in India. He noted the frons of S. litura is dark brown to black, the spiracle of A7 is two thirds as large as
the one on A8, and SD1 is dorsad of the spiracle on the abdominal segments. The labral notch is wide but shallow, the setae are short, the skin is
smooth, and the spiracles are black. Later, Gardner (1947) placed S. litura in his "Division A, group A2," using some of the above characters.
Members of group A2 appear to have a relatively unspecialized morphology; perhaps the most significant characters are the short setae, position
of SD1 dorsad of the spiracle on A8, a bisetose SV group on A1, a trisetose SV group on A2, stemmata 1 and 2 closely spaced and the frontal
pores in line with the F1 setae. The spinneret of S. litura has two projections and an elongate rounded apical lobe (Gardner 1947: fig. 16).
Chu et al. (1965: plate 5: fig. 80) illustrated the larva of S. litura from China. The P setae are evenly spaced, the labrum has a developed
notch, the apex of the spinneret has an elongated round apical lobe, and his illustration of the mandible shows five teeth with a few minute serrations on the
cutting margin. A black patch on the abdominal segments surrounds both the spiracle and a lighter spot at the posterior margin.
Mochida (1973) compared twenty larvae each of S. litura from Japan to S. littoralis from Egypt. The basal margin of the labrum is convex in
S. litura but linear in S. littoralis. The pattern of coloration around the abdominal spiracles was better developed in S. litura than to
S. littoralis. Other features of the head, mouthparts and thoracic claws were considered "characteristic" of each species but they were not
enumerated by Mochida (1973). The figures do show some major differences. Spodoptera litura has the MD setae in a straight line on the head, P2 almost dorsad of P1,
a rectangular mandible, and the apex of the spinneret rounded. This differs from S. littoralis, which has the MD setae in a curved line, P2 more widely spaced
from each other than P1, a square shaped mandible, and the apex of the spinneret pointed. Both species have four setae on the prothoracic shield.
Weisman (1974) defined a "Prodenia group" that included S. litura and other species of Spodoptera. He mentioned a broad and flat spinneret,
almost equal in length to the labial palpus, with a middorsal groove and two small projections at the apex. The SD setae are off the prothoracic
shield and SD1 of the mesothorax [and metathorax] are connected to the associated tonofibrillary platelet by a minute sclerotized bar.
The mandible has no retinaculum.
Aitkenhead et al. (1974) thought the markings on A1 of S. litura extended more laterally than in S. littoralis but otherwise two species could
not be told apart in the field. Brown and Cantrell (1978) illustrated the mandible of S. litura and noted the presence of white spots on the dorsum.
Beardsley (1982) published a key to Hawaiian noctuids. After eliminating unrelated species, he separated S. litura from Elaphria and other
Spodoptera in Hawaii by the spacing of stemmata 3 and 4, spiracle size and color, the lack of dorsal round spots on A2, and the relatively broad
spinneret. The description of S. litura by Thakar and Srivastva (1983) included a short text and diagrammatic figures. There was no diagnosis.
Bejakovich and Dugdale (1998: 16, 17, 52) defined S. mauritia and S. litura by having L2 below the level of the spiracle on A8; the D, SD and L setae
with a slightly truncate or expanded apex; presence of "eyespots" (white spots) on the mesothorax and metathorax and a swollen area at D2 on the last
two thoracic segments compare to the prothorax. They recognized two color forms of S. litura in New Zealand interceptions. Both have a white spot
on a dark "sclerite" [patch] posterodorsad of the abdominal spiracles. These markings may be "vivid" (S. litura from Australia) or "drab"
(S. litura from the eastern Pacific).
Pogue (2002) added that the distance between P2 is greater than that separating the P1 setae, the mandible has teeth, the front extends about 1/2 to 3/4 the
distance to the epicranial notch and there are about 127 crochets on one side of the body. He also recognized two color forms. Important color characters
include the dorsal segmental triangles of A7 and A8 being larger than those on A1-6, all with a small white spot at their apex, and the presence of a
white or light spot caudal to the spiracle.
Passoa (2011) compared S. litura and S. littoralis to other North American Spodoptera as part of a screening key to allow more rapid recognition of
these exotic species if ever introduced to North America. For typical specimens of S. litura, the dorsal triangle of A1 is larger than the triangle on
A6, the spiracular line may be interrupted on A1 but there is no loss of intensity on the thorax compared to the abdomen and dorsal spots are often
present on T2 and T3. The ground color varies from green to brown, a middorsal line may be present and the abdominal triangular markings have a white
spot at the apex.
The unspecialized morphology of S. litura is shown by its location on the last couplet of Beardsley (1982), and both keys by Gardner (1941, 1947),
only after all the other species are eliminated. From the above studies, we suggest the following morphological characters: spinneret with two
projections and an elongate rounded apical lobe; mandible with four scissorial teeth and no retinaculum; SD1 on T2 and T3 connected to the associated
tonofibrillary platelet by a minute sclerotized bar; SV group bisetose on A1; SD1 dorsad of the spiracle on A8 and the body setae short, most not much
longer than the vertical height of the 8th abdominal spiracle. Passoa (1991) listed nine general characters to separate Spodoptera from other economic
noctuids in Honduras, a few of these not mentioned above are applicable to S. litura, for example, four abdominal prolegs present (larva not a looper),
lateral spot often present on first abdominal segment (this spot is a practical method of recognizing Spodoptera larvae in the field), hypopharyngeal
complex with coarsely spined posterior portion lacking a dense brush of stout bristles, cuticle smooth under low magnification, and head with
an inverted "Y" because adfrontal areas are outlined in white (see Wagner et al. 2011). There appears to be variation in the spinneret of S. litura as
the figures of Chu et al. (1965) and Mochida (1973) do not match, perhaps the former being more typical. There also seems to be variation in the spacing
of the P setae, the labral notch and the shape of the mandible according to the available illustrations. With our limited material, we could not confirm
that the D, SD and L setae of S. litura have a slightly truncate or expanded apex as mentioned by Bejakovich and Dugdale (1998).
There is a long ridgelike blade on the hypopharyngeal complex and SD1 is hairlike on A9.
The following color characters are most important for recognizing S. litura: middorsal line often present and conspicuous; dorsal triangles absent on all
segments, present on A1 to A8, only A1 and A8 or just A8; the dorsal triangle of A1 usually much larger than that of A6; the spiracular line, if interrupted
on A1, then equal in intensity on both the thorax and abdomen; abdominal spiracles with a dorsal dark spot enclosing a smaller white spot posterior to the
spiracle, and apex of dorsal triangles with a small apical white spot. These patterns are visible on living and usually preserved larvae.
There are both larval and adult color differences for S. litura across its range (Bejakovich and Dugdale 1998, Pogue 2002). Both the "drab" and "vivid"
larval color forms occur together in Japan (Komai et al. 2011: plate 236). The significance of morphological or color variation of S. litura is not known.
We have chosen the most common states in both morphology and color based on United States port interceptions. Exceptions to the above diagnosis are
known and more may be expected. Bejakovich and Dugdale (1998) and Mohn (2001) illustrated examples of a S. litura with a pale abdominal dorsum and only
traces of a spiracular stripe or abdominal triangles. In other cases
(Example Here),
the dorsal triangles are well developed and all are almost equal in size.
Identification authority (Detailed)
Because the genus Spodoptera is hard to define as larvae, care must be taken to be sure the specimen is in this genus. The thoracic bar on SD1
occurs in many noctuids, it is not an indication of Spodoptera by itself. Morphological characters were described in detail above to help with
doubtful cases. Both the morphology and color must match Spodoptera. If either is not correct, it is better to stay at family Noctuidae.
Accurate identification of S. litura involves origin, morphology and color pattern. Because this pest is polyphagous, the hostplant offers
few clues. The one exception is S. litura on orchids from Thailand.
There have been interceptions of Spodoptera on orchids from Thailand for many years, going back to at least to the 1970's (Ford 1988: 6).
Some of these are S. exigua, easily recognized by the characters given in our data sheet. Because orchids are not a field host for S. litura,
there was doubt about this association (Ford 1988). However, the Netherlands Plant Protection Organization has reared S. litura from Thailand
orchids several times from 2005-2007 (M. van der Straten, pers. comm.), thus confirming this pathway. Because there are still only two easily
separated Spodoptera on orchids from Thailand, even young larvae of S. litura (second to last instar) can be named. This is the rare exception,
otherwise, larvae of Spodoptera with an unknown origin, or those young enough to have a swollen thorax, should only be identified to genus.
Typically, the forms of S. litura can be summarized as follows. Early instars have a swollen thorax and usually either a band or a large
black spot on A1. Heavily marked later instars have white spots on the mesothorax and metathorax, a middorsal stripe, dorsal triangles on all
the abdominal segments with apical white spot and abdominal spiracles with a dorsal dark spot enclosing a smaller white spot posterior to the
spiracle. Drab or poorly marked forms of S. litura will lack the thoracic spots, dorsal triangles and either or both of the spots near the
spiracle. In these cases, origin may be the only way to separate them from S. littoralis pending confirmation of morphological characters
mentioned by Mochida (1973). Spodoptera litura can be identified from India west throughout Asia; S. littoralis from about Israel east to
northern Africa and Europe. Doubtful specimens from Israel to India can be left at genus. The presence of a middorsal stripe in S. litura
was considered distinctive (EPPO data sheet), but this character is also variable. Although S. litura does not occur in Europe naturally,
it is common in cut flowers from the Netherlands.
NOCTUIDAE - Spodoptera litura (Fabricius)
Origin records
Spodoptera litura has been intercepted from the following locations:
China, Hawaii, Hong Kong (?), India, Israel, Japan, Malaysia, Netherlands, New Zealand, Philippines,
Singapore, Taiwan, Thailand, Viet Nam
Although S. litura does not occur in Europe naturally, it is common in cut flowers from the Netherlands.
Host records
Spodoptera litura has been intercepted on the following hosts:
Actinidia sp., Alocasia sp., Anemone sp., Anethum graveolens, Arachnis sp., Aranda sp., Aranthera sp.,
Ascocenda sp., Begonia sp., Brassica sp., Callistephus chinensis, Capsicum sp.,
Chrysanthemum sp., Citrus sp., Cleome sp., Cymbidium sp., Dendrobium sp., Dischidia pectenoides,
Echinodorus sp., Euphorbia lactea, Euphorbia sp., Gardenia sp., Gomphrena globosa, Jasminum sp.,
Lactuca sativa, Lisianthus sp., Lotus sp., Ludwigia sp., Mokara sp., Nelumbo nucifera, Nelumbo sp.,
Nymphaea sp., Ocimum sanctum, Ocimum sp., Oncidium aureum, Oncidium basilicum, Oncidium bicallosum,
Oncidium sp., Orchidaceae, Origanum sp., Origanum vulgare, Pelargonium sp.,
Philodendron sp., Platycerium sp., Rosa sp., Rotala sp., Sageretia thea, Solanum sp.,
Tagetes sp., Thymus sp., Vanda sp., Zantedeschia sp.
NOCTUIDAE - Spodoptera litura (Fabricius)
Setal map
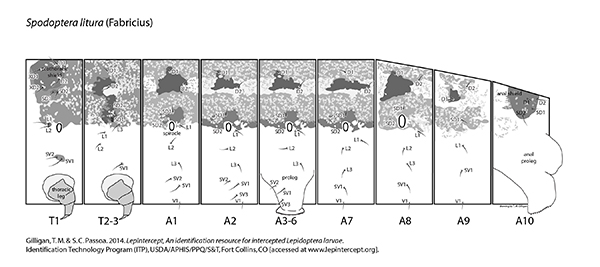 Spodoptera litura setal map Spodoptera litura setal map
<< Previous fact sheet Next fact sheet >>
 Fig. 1: Late instar, lateral view
Fig. 1: Late instar, lateral view |
 Fig. 2: Late instar, lateral view
Fig. 2: Late instar, lateral view |
 Fig. 3: Early to mid-instar, lateral view
Fig. 3: Early to mid-instar, lateral view |
 Fig. 4: Early instar, lateral view
Fig. 4: Early instar, lateral view |
 Fig. 5: Late instar, dorsal view
Fig. 5: Late instar, dorsal view |
 Fig. 6: Late instar thorax, lateral view
Fig. 6: Late instar thorax, lateral view |
 Fig. 7: Early instar, lateral view
Fig. 7: Early instar |

Fig. 8: Crochets |
 Fig. 9: Head with adfrontal area outlined in white forming an inverted "Y"
Fig. 9: "Y" on head |

Fig. 10: Head |
 Fig. 11: Hypopharyngeal complex, lateral view
Fig. 11: Hypo. complex |

Fig. 12: Mandible |
 Fig. 13: Hypopharyngeal complex, dorsal view
Fig. 13: Hypopharyngeal complex, dorsal view |
|
|
|
|
|