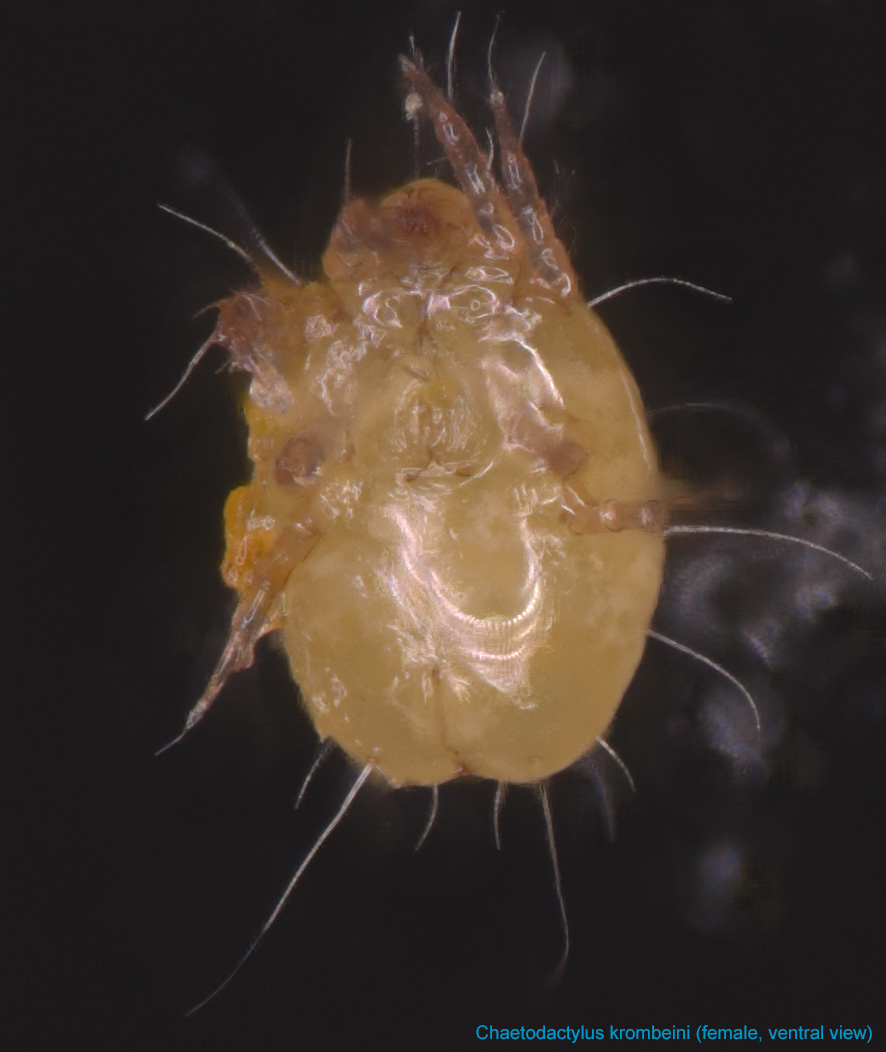
Fig. 9. Chaetodactylus krombeini female, ventral view; FL photo by Ron Ochoa & Gary Bauchan, USDA-ARS.
Fig. 9. Chaetodactylus krombeini female, ventral view; FL photo by Ron Ochoa & Gary Bauchan, USDA-ARS.

Fig. 10. Chaetodactylus krombeini male, ventral view; FL photo by Ron Ochoa & Gary Bauchan, USDA-ARS.
Fig. 10. Chaetodactylus krombeini male, ventral view; FL photo by Ron Ochoa & Gary Bauchan, USDA-ARS.
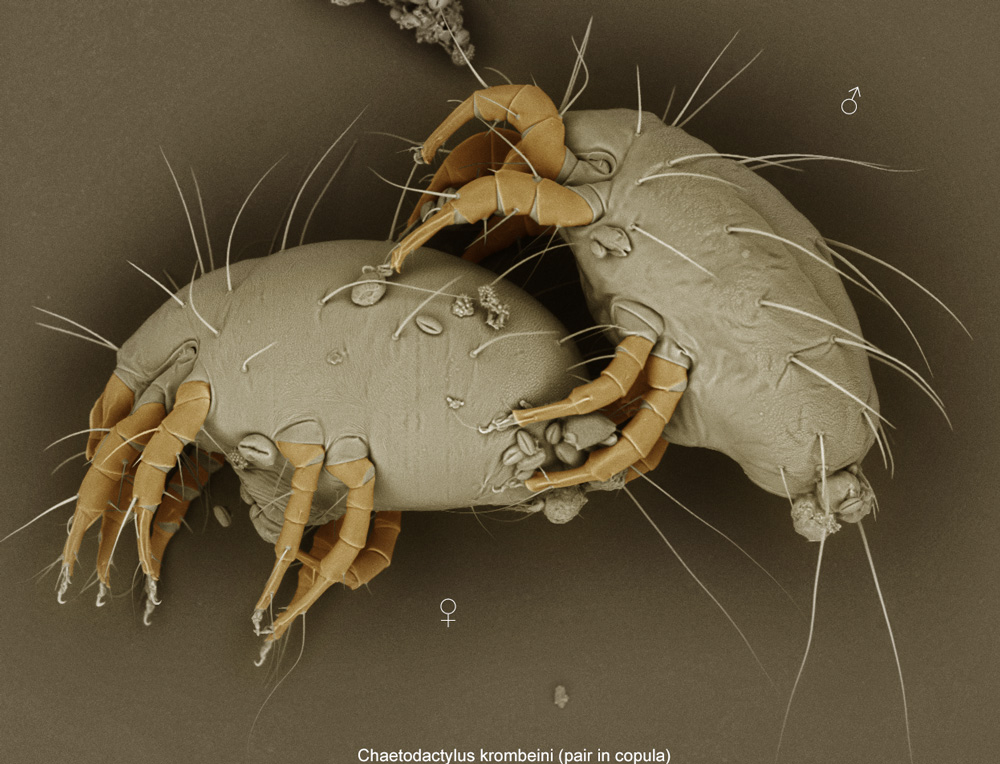
Fig. 11. Chaetodactylus krombeini pair in copula; LT-SEM photo by Ron Ochoa & Gary Bauchan, USDA-ARS.
Fig. 11. Chaetodactylus krombeini pair in copula; LT-SEM photo by Ron Ochoa & Gary Bauchan, USDA-ARS.
.jpg)
Fig. 12. Chaetodactylus krombeini female lateral propodosoma showing enlarged supracoxal sclerite. This trait is diagnostic for the family Chaetodactylidae; LT-SEM photo by Ron Ochoa & Gary Bauchan, USDA-ARS.
Fig. 12. Chaetodactylus krombeini female lateral propodosoma showing enlarged supracoxal sclerite. This trait is diagnostic for the family Chaetodactylidae; LT-SEM photo by Ron Ochoa & Gary Bauchan, USDA-ARS.
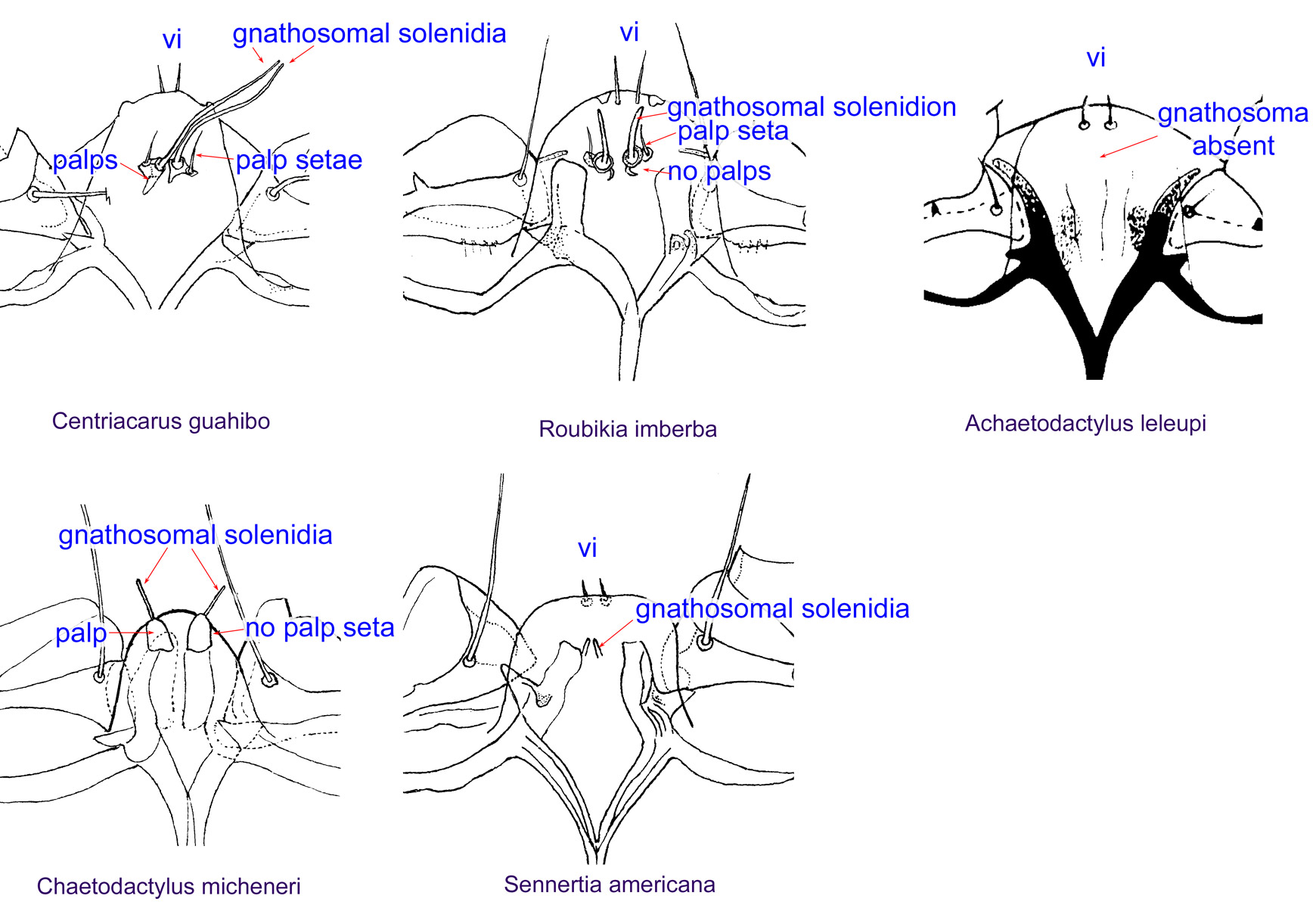
Fig. 13. Gnathosomal morphology is a key character separating genera of the family Chaetodactylidae (Centriacarus, Roubikia, Achaetodactylus, Chaetodactylus, and Sennertia) based on phoretic deutonymphs. In particular, Chaetodactylus can be distinguished by having palps and gnathosomal solenidia present and palp setae absent; drawing of Achaetodactylus leleupi courtesy of Belgian GTI Focal Point 2009, http://www.taxonomy.be.
Fig. 13. Gnathosomal morphology is a key character separating genera of the family Chaetodactylidae (Centriacarus, Roubikia, Achaetodactylus, Chaetodactylus, and Sennertia) based on phoretic deutonymphs. In particular, Chaetodactylus can be distinguished by having palps and gnathosomal solenidia present and palp setae absent; drawing of Achaetodactylus leleupi courtesy of Belgian GTI Focal Point 2009, http://www.taxonomy.be.
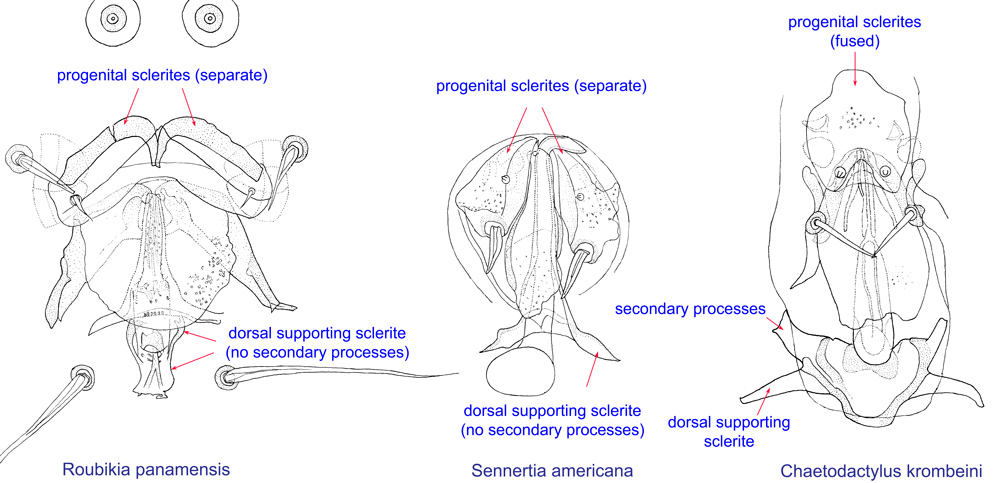
Fig. 14. External genital appartus of males of Roubikia, Sennertia, and Chaetodactylus, showing key diagnostic features of Chaetodactylus: fused progenital sclerites and dorsal supporting sclerite with secondary processes.
Fig. 14. External genital appartus of males of Roubikia, Sennertia, and Chaetodactylus, showing key diagnostic features of Chaetodactylus: fused progenital sclerites and dorsal supporting sclerite with secondary processes.
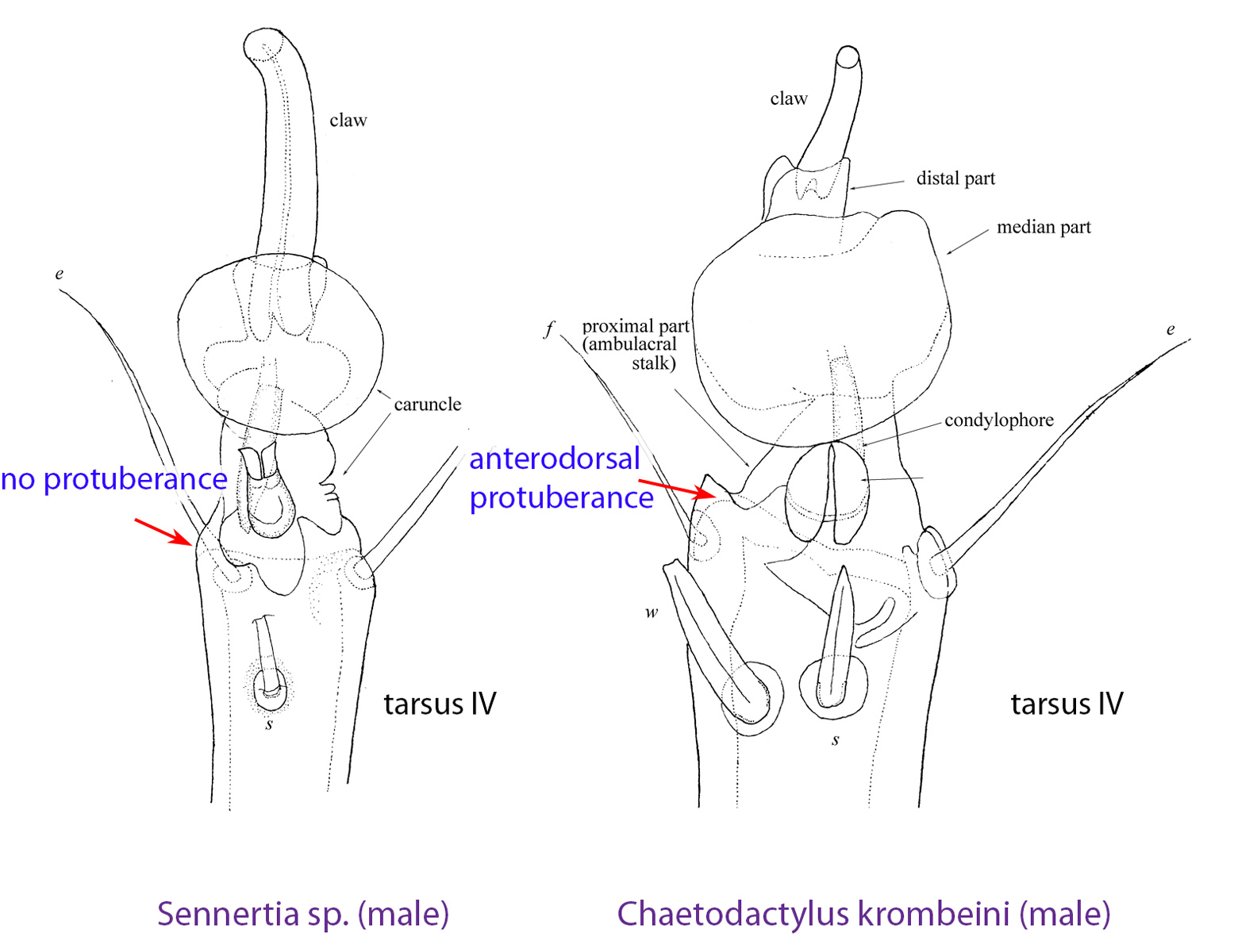
Fig. 15. The presence of an anterodorsal protuberance on tarsi I-IV of males is a unique feature of Chaetodactylus. For comparison, tarsus IV of Sennertia sp. is also shown.
Fig. 15. The presence of an anterodorsal protuberance on tarsi I-IV of males is a unique feature of Chaetodactylus. For comparison, tarsus IV of Sennertia sp. is also shown.

Fig. 16. Chaetodactylus krombeini phoretic deutonymphs dispersing on the blue orchard bee Osmia lignaria.
Fig. 16. Chaetodactylus krombeini phoretic deutonymphs dispersing on the blue orchard bee Osmia lignaria.

Fig. 17. Phoretic deutonymphs of Tortonia smitsvanburgsti and Chaetodactylus anthidii on bee Rhodanthidium sticticum; photo by Lindsey Seastone & Laura Hartmann, ITP.
Fig. 17. Phoretic deutonymphs of Tortonia smitsvanburgsti and Chaetodactylus anthidii on bee Rhodanthidium sticticum; photo by Lindsey Seastone & Laura Hartmann, ITP.

Fig. 18. Chaetodactylus krombeini mites in a brood cell of the blue orchard bee Osmia lignaria; photo by USDA-ARS.
Fig. 18. Chaetodactylus krombeini mites in a brood cell of the blue orchard bee Osmia lignaria; photo by USDA-ARS.

Fig. 4. Females of Chelacheles temoak phoretic on bee Diadasia sphaeralcearum (bee photographed after mites were removed).
Fig. 4. Females of Chelacheles temoak phoretic on bee Diadasia sphaeralcearum (bee photographed after mites were removed).
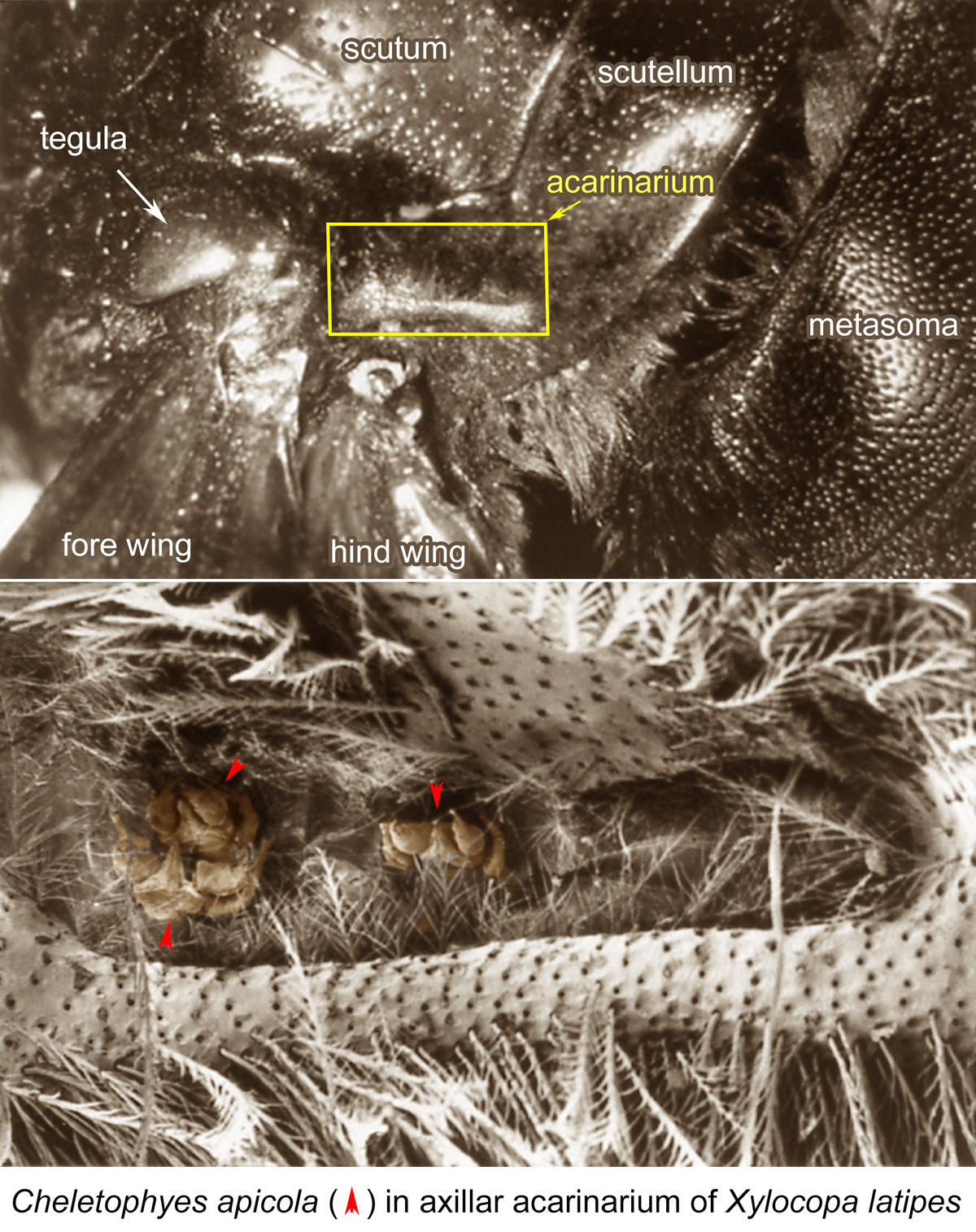
Fig. 6. Cheletophyes apicola phoretic females in axillar acarinarium of Xylocopa latipes; SEM photo by Barry OConnor, University of Michigan.
Fig. 6. Cheletophyes apicola phoretic females in axillar acarinarium of Xylocopa latipes; SEM photo by Barry OConnor, University of Michigan.